Varithena™
What is Varithena™?
 Varithena™ is the prescription medicine polidocanol injectable foam 1%. It is used to treat varicose veins caused by problems with the great saphenous vein and other related veins in the leg’s superficial system. Varithena™ improves symptoms related to or caused by varicose veins and the appearances of varicose veins.
Varithena™ is the prescription medicine polidocanol injectable foam 1%. It is used to treat varicose veins caused by problems with the great saphenous vein and other related veins in the leg’s superficial system. Varithena™ improves symptoms related to or caused by varicose veins and the appearances of varicose veins.
Is Varithena™ safe?
Varithena™ has been studied in multiple clinical trials for safety. Varithena™-treated patients were compared with a group of patients treated with placebo and Varithena™ has an excellent safety profile. Treatment-adverse reactions were 3% higher with the Varithena™-treated patients compared with the placebo-treated patients.
What are the benefits of Varithena™?
Varithena™ is administered through an IV or a needle and because the chemically formulated foam travels along the course of the diseased veins and works from the inside of the vein without any heat energy, patients do not have to have tumescent anesthesia (a series of needles to inject numbing medicine around the culprit vein). The procedure is quick and many patients do not have any sensation of pain or discomfort with injection of the foam.
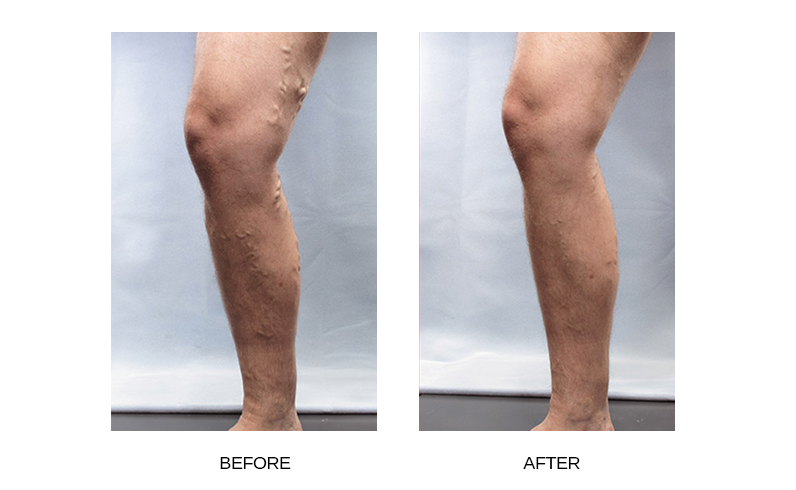
What results are expected with Varithena™?
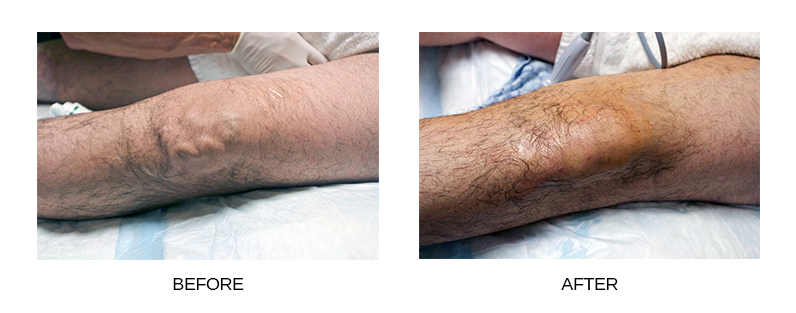
In clinical trials, Varithena™ reduced patients’ varicose vein symptoms including heaviness, achiness, swelling, throbbing, and itching. Varithena™ also improved vein appearance in assessments by both patients and doctors.
What can I expect during treatments with Varithena™?
Varithena™ treatments are usually out-patient, painless procedures that take less than 60 minutes. Varithena™ is administered through a simple IV into your malfunctioning vein and its branches. The foam fills the inside of the vein and the vein reacts to the foam by collapsing. Blood flow shifts to healthier veins nearby. Since there are no incisions, most patients experience little discomfort although the injection site is usually numbed and this can be uncomfortable for some patients.
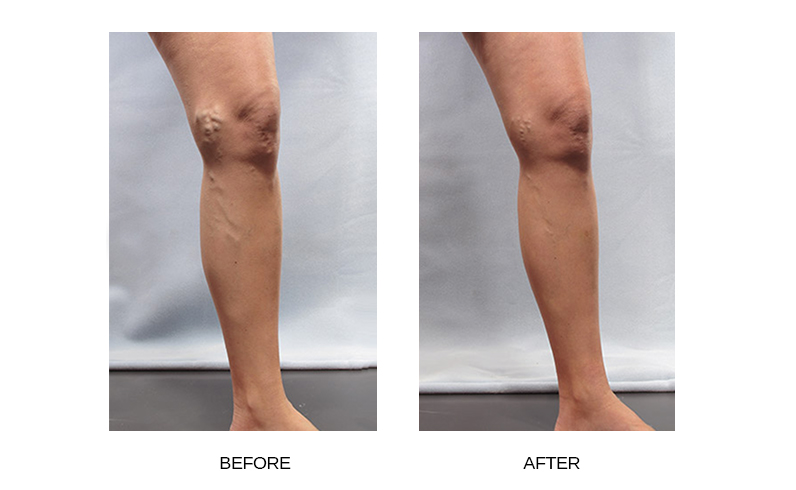
What aftercare is recommended following Varithena™ treatment?
Post-treatment compression is required for the first 48 hours followed by a 2-week course of gradient compression stockings. Once your compression is applied post-procedure, you will be asked to walk for at least 10 minutes under supervision. You should walk at least 30 minutes daily and avoid long periods of inactivity and any weight-lifting activities for the next month. No heavy exercise is recommended during the first week after treatment.
What are the possible side effects associated with Varithena™?
The most common side effect with Varithena™ is leg pain or discomfort. In the majority of cases, leg pain stops within one week. The other common side effects are injections site bruising or pain. Discoloration of the treated vein may occur. Rarely, Varithena causes a life-threatening allergic reaction.
Am I a candidate for Varithena™?
Varithena™ can be safe for most patients. Patients who are allergic to polidocanol or have blood clots in their veins are not candidates for Varithena™. Varithena™ will not be administered during pregnancy. If you have any of the following conditions, you may not be a suitable candidate for Varithena™:
- Peripheral arterial disease
- Reduced mobility
- A history of blood clots in the veins or the lungs
- A history of major surgery in the past 3 months
- A history of a prolonged hospitalization in the past 1 year
What does Varithena™ cost?
Please check out our PRICING PAGE.
Schedule a Consultation Today
We encourage you to schedule a consultation today. During our meeting, we will advise on whether or not you’re an ideal candidate for this procedure, as well as compose a custom treatment plan designed to achieve the smooth, clear and vein-free skin you’ve always wanted. Call, text or fax the office today and one of our seasoned staff members will be able to assist you.
Call | Text | Fax
Veins 101: Why do Varicose Veins itch?
We know how frustrating varicose veins can be... Not only are they unsightly, but they ITCH. Why do they itch?!?! The reason is a condition associated with varicose veins called Venous Stasis Dermatitis. This condition is caused by blood building up in the veins and...
Veins 101: How are Varicose Veins Treated?
One of the most common questions people with Varicose Veins ask is: How do I get rid of them? At Totality, we offer several treatment modalities to get rid of those persistent Varicose Veins. Previously, Varicose Veins were physically cut out of the body, resulting in...
Veins 101: Vaginal Varicose Veins
Vaginal Varicose Veins… What a nuisance! Unfortunately, pregnant individuals are most at-risk for developing them. Vaginal Varicose Veins are very normal, and are referred to as Vulvar Varicosities, since they don't actually impact the vaginal canal and only the...